5 tips to take care of your mental health during COVID-19
March 30, 2021
Mental Health Week
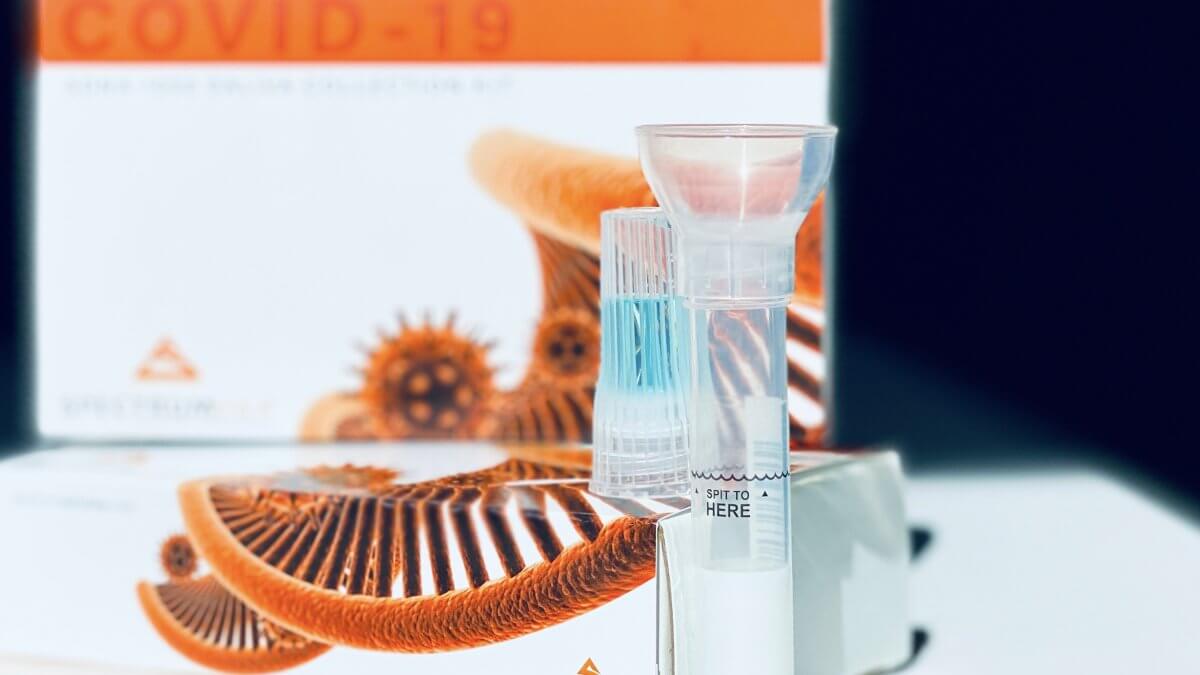
May 7, 2021TORONTO, May 4, 2021 /CNW/ – A saliva collection kit that uses a pain-free method to test for COVID-19 is being distributed in Canada for the first time by RxBIO Corp., a leading medical device distribution company.
Spectrum Solutions’ SDNA saliva collection kit offers a comfortable and more accurate alternative to the painful nasal swab test method for COVID-19. Approved by Health Canada under Interim Order in January 2021, Spectrum’s SDNA saliva collection kit is the first saliva-based system to receive Emergency Use Authorization for COVID-19 testing by the U.S. Food and Drug Administration.
The launch of Spectrum’s SDNA-2000 and SDNA-3000 Saliva Collection Devices in Canada means Canadians have access to an effective saliva-based COVID-19 test option, with 99.998 percent accuracy. The device is so reliable that Major League Baseball uses Spectrum’s SDNA-1000 saliva collection kits to test all players and staff.
To read the full press release, visit: https://www.newswire.ca/news-releases/rxbio-brings-covid-19-saliva-collection-kit-with-99-998-accuracy-to-canadians-888246483.html
or: mediacoverage.com for more details!